Next-Gen
Oncology
CRO Services
Oncology drug development has changed and we have changed with it. Next-generation targeted therapies require a next-generation oncology CRO with the aligned services to navigate and execute complex clinical trials in an efficient and comprehensive way.
Are you looking for a best-in-class partner to expedite your targeted oncology therapy? Precision for Medicine was purpose-built to meet the needs of personalized medicine drug development and commercialization.
Oncology CRO services spanning discovery, development and commercialization
Precision was purpose-built as an oncology CRO to support the unique demands of personalized medicine drug development, specifically in the areas of oncology and hematology.
Precision Convergence is our suite of aligned oncology CRO services, focused on bringing you world-class teams and technologies to support your research and development requirements. From discovery and clinical development, through manufacturing, commercialization, and beyond, our team is globally positioned to help you deliver your oncology therapy at scale.
Oncology CRO services delivered by exceptionally
dedicated clinical teams
Recent oncology drug approvals supported by Precision teams
(within past 3 years)

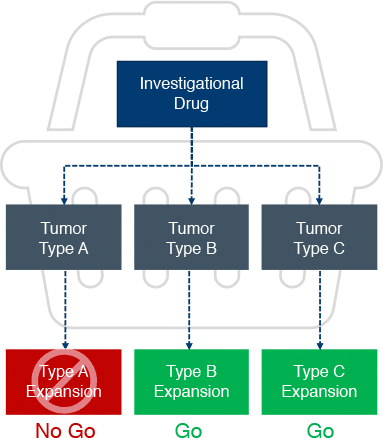
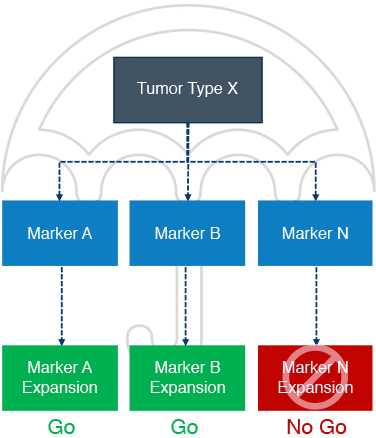
Experts in basket & umbrella trials for oncology early phase development
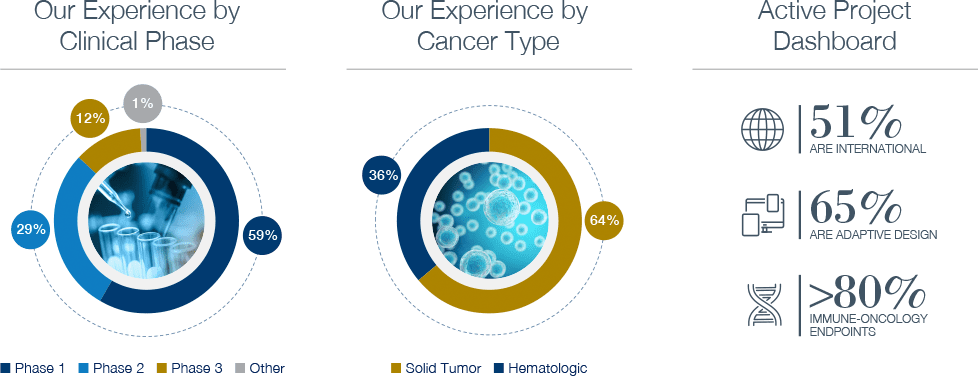
As an oncology contract research organization, we excel at running complex trials such as basket and umbrella trials with adaptive design, master protocols with multiple treatment cohorts, and biomarker-driven trials with intricate sample collection. Our team of clinical trial design experts is ready to assist on your next trial.
Biomarker assay development and validation through to companion diagnostics
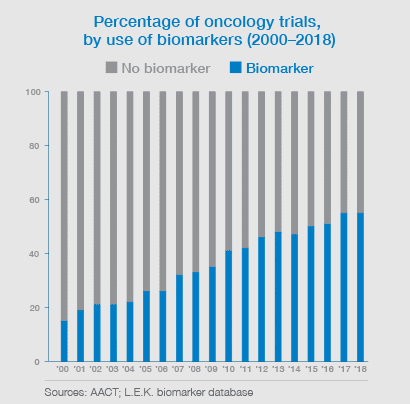
Biomarker use in Oncology clinical trials for the industry is now over 55% of all studies. Biomarker validation is critical to your drug and clinical program’s success. Delivering new biomarker tests to support clinical trials and personalized therapy is our passion. Work with the leaders in biomarker development and validation at Precision for Medicine.
Oncology Expertise in Biomarker Development including:
- Liquid biopsy-based biomarkers: ctDNA or CTC
- Solid tumor biomarkers: IHC, multiplex immunofluorescence
- Immune monitoring biomarkers: Flow cytometry, epigenetic cell phenotyping
Developing a companion diagnostic for your clinical program? Precision for Medicine has over 15 years of experience as an oncology CRO in guiding sponsors down the companion diagnostic pathway.
Learn More – Precision Companion Diagnostics

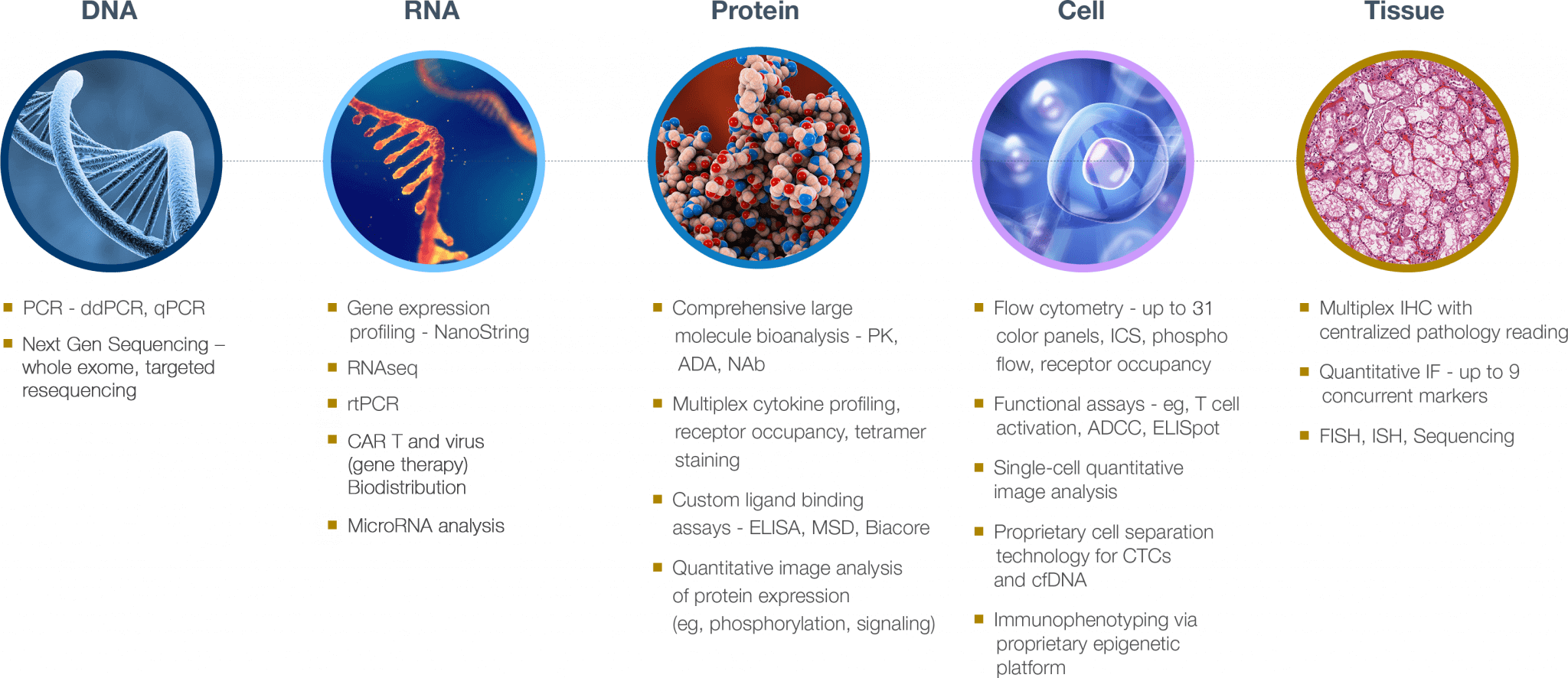
Oncology CRO expertise in developing and validating biomarker assays, for any tissue type
Proprietary technologies empowering Precision’s oncology CRO services
Part of what makes Precision unique is our history of scientific innovation as an oncology CRO. Not only do we combine state-of-the-art approaches to maximize the information each tumor provides, we create proprietary technologies that solve oncology and immuno-oncology clinical research study challenges.
Proprietary specialty labs services
Epiontis ID® – Precision’s patented Epiontis ID technology is a robust, flexible, and cost-effective platform for immune cell phenotyping based on epigenetic cell counting. With minimal sample processing requirements that simplifies global trial logistics and robust, reproducible results that can be compared between sites and studies, Epiontis ID is an ideal solution for immune monitoring in both early-stage and late-stage clinical trials. Learn more about Epiontis ID
ApoStream® – an antibody-free method for capturing and enriching circulating tumor cells (CTCs) as well as other rare cell types from liquid biopsies. When isolated from blood, CTCs are a more accessible source of information about the primary cancer than a surgical biopsy. Learn more about ApoStream
Managing & analyzing diverse biological data via QuartzBio® software solutions
Modern biomarker-driven programs generate massive volumes of data throughout the course of development. Just as Electronic Data Capture (EDC) technology revolutionized our industry’s approach to clinical data management, QuartzBio revolutionizes biomarker operations, data management, and analytics.
virtual Sample Inventory Management (vSIM) – Oncology trials today are collecting more and more biological data samples from patients. QuartzBio’s solution for virtual Sample Inventory Management centralizes and harmonizes sample data and metadata from all central labs, specialty labs, biorepositories, and EDC. Learn more about virtual Sample Inventory Management
enterprise Biomarker Data Management (eBDM) – Enable real-time data access and inform critical decisions through integration and centralization of biomarker data, clinical data, and publicly available datasets into one central, accessible database with a suite of visualization, analysis and reporting tools. Learn more about enterprise Biomarker Data Management

“We have been delighted with the experience level and work product from the entire project team – they feel like an extension of our internal team.”
Director, Clinical Operations
Midsize Pharma
Companion diagnostics development & consulting oncology CRO services
Are you planning a companion diagnostic? Precision’s Companion Diagnostics team can guide and support your development, regulatory, and commercialization journey. The route to market for a companion diagnostic is complex and must be well planned to ensure that the test and drug come to market together, with the best possible patient access.
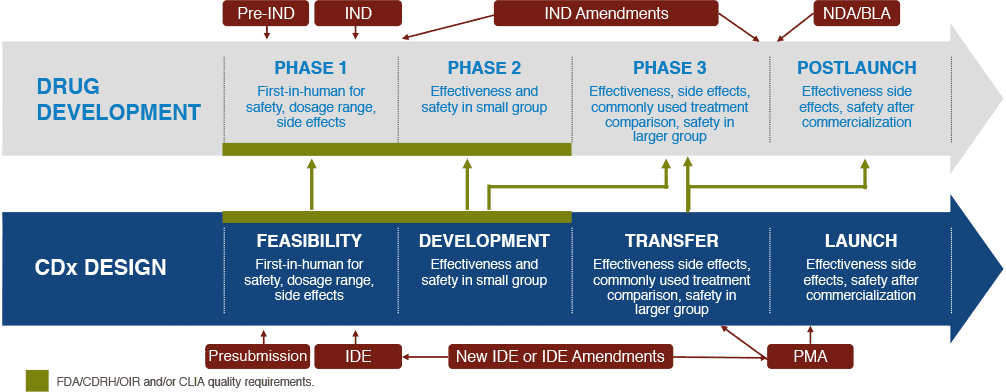
Leveraging site relationships while gaining deeper data insights from every patient enrolled
Precision oncology site network
Working with a CRO partner that knows and has relationships with the Oncology Investigator community matters. A focused Oncology CRO, we know selecting the right sites for your clinical trial can predetermine many factors like completing enrollment on time or having quality in your data. Having supported oncology clinical trials for 20+ years, Precision has built strong relationships and established the Precision Oncology Site Network (OSN).
Precision OSN brings our sponsors access to 80+ Oncology Centers from around the world that are preidentified, prequalified, and have extensive infrastructure, expertise and, most importantly, access to the patient populations you need for your trials.
Learn more about our Precision Oncology Site Network HERE.
Deeper insights provided by Precision CSAI
Our Clinical Science Analytics & Insights team is part of our integrated and cross-functional data review process, which solves the typical gap between standard data review and application of oncology clinical research standards, such as RECIST or iRECIST 1.1 and CTCAE criteria.
Every data point and every patient matters in your trial and applying a process that implements the right functional reviews at the right time to ensure site adherence and consistency allowing for data driven decisions throughout the study is critical to ensure high quality data. The CSAI team leverages their Oncology expertise and knowledge to make certain your data is examined holistically with indication specific standards applied and adhered to across your dataset.
CSAI leverages JReview Cloud, SAS, and Precision’s SmartPatient profiles to support data visualization and remote data review by your team and ours.
Interested in learning more about our CSAI team and service for your next oncology trial? Speak with our team, and we would be delighted to provide a quote for this critical and differentiating service.
YEARS FASTER AVERAGE APPROVAL TIME FOR PRECISION MEDICINE
Precision Convergence, the combined power of trials, labs and data sciences to drive faster clinical development
What sets us apart is the way we integrate clinical trial execution with deep scientific knowledge, laboratory expertise, and advanced data sciences. This is Precision Convergence: maximizing insights into patient biology and accelerating the pace of scientific discovery and approval.