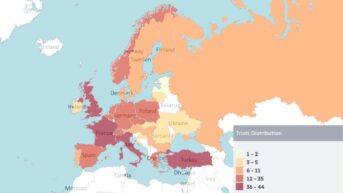
Neurology and CNS Clinical Trials Development
Due to the complexity of neurology and CNS clinical trials, successful execution requires careful planning, the right technology, bioinformatics expertise, and a disciplined clinical approach. We support neurology therapeutic development with over 25 years of experience designing and executing neurology and CNS clinical trials, particularly in the orphan disease space.
At Precision, we incorporate advances in neurology biomarkers and biomarker detection technology to build a robust multi-parameter biomarker strategy for therapeutic area development.
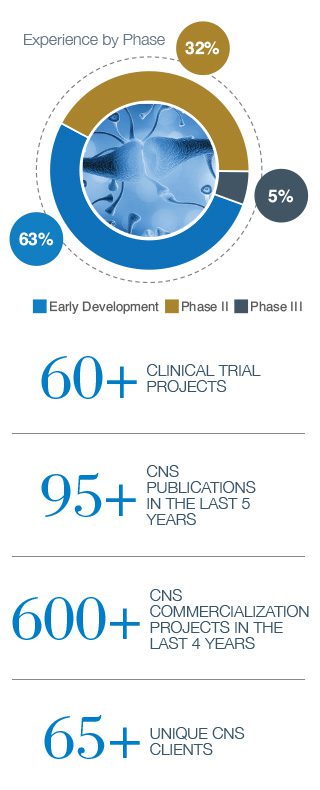

Our proprietary and advanced biomarker detection platforms power neuroscience and CNS clinical research
In addition to many standard biomarker detection and analysis platforms (eg, flow cytometry, ELISA), Precision also utilizes state-of-the art and proprietary technology platforms to enable detection of the most relevant and actionable neurology biomarkers.
- Quanterix® SIMOA® is a platform that detects low-level, blood-based, CNS-derived biomarkers to monitor neuronal pathophysiology including proteinopathies.
- Epiontis ID®, our proprietary epigenetic qPCR-based technology for fluid-based immune cell monitoring and immunophenotyping, has several advantages over flow cytometry and has been successfully utilized to enable clinical studies in neurology, including in multiple sclerosis.
- QuartzBio®, proprietary solutions that enable multi-parameter, multi-omic approaches to biomarker discovery and development, can harmonize and support the analysis of biomarker data from multiple sources and assay types while utilizing computational biology approaches to extract insight and value – including the identification of biomarker patterns correlating with disease subtypes and progression.
- Neuron-Derived Exosomes (NDEs), which are isolated from patient blood, provide a non-invasive means to monitor neuronal physiology and pathophysiology. NDEs are amenable to standard downstream biochemical assays to facilitate the identification and validation of novel diagnostic, prognostic, and predictive biomarkers.
Over 25 years of experience designing and executing neurology & CNS clinical trials
As a CNS CRO, Precision delivers end-to-end, biomarker-driven clinical execution (rapid study startup, integrated biomarker strategies, and full-service, complex, global CNS clinical trials) to accelerate the path to market for emerging and mid-stage biopharma companies in the United States, European Union, and Asia-Pacific. What makes Precision unique are the scientists, clinicians, and operational experts dedicated to biomarker-driven clinical development.
Over 60 CNS clinical research studies, including those on:
- Alzheimer’s disease
- Parkinson’s disease
- Rett syndrome
- Insomnia
- Pain
Expertise in supporting gene therapy–based neuroscience therapeutic areas
Successfully bringing a gene therapy to market requires a deep understanding not only of the science, but also of the specific regulatory needs unique to gene therapies. Precision provides end-to-end solutions to support development of gene therapy products.
- Leadership in regulatory strategy
- Gene therapy–specific assay design and execution
- Specialization in immunogenicity studies
- Excellence in rare and orphan disease trials (awarded Best Orphan Drug CRO for 2 consecutive years)
- Expertise to effectively coordinate both a therapeutic and companion diagnostic strategy in parallel

Insights into CNS Clinical Development
CNS & Neuroscience CRO Services, Spanning Discovery, Development, and Commercialization
At Precision, complexity is our playground. We specialize in managing the unique demands of personalized medicine for the most sophisticated areas of our bodies. Our suite of aligned services combines world-class teams and technologies to support your CNS and neuroscience clinical research and development requirements from discovery and clinical development through manufacturing, commercialization, and beyond. Our global footprint ensures we can support your neuroscience research end-to-end.
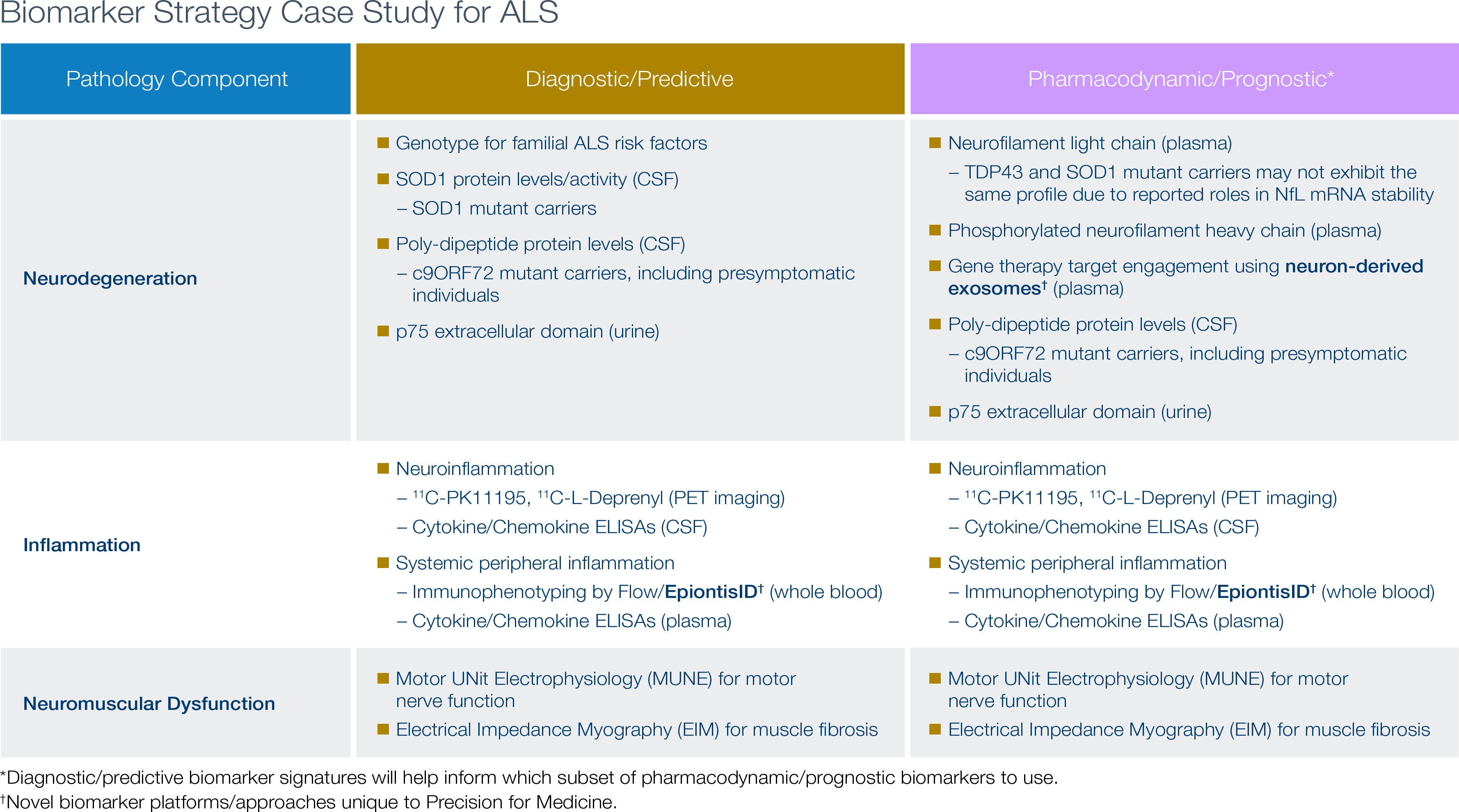
Case Study: Development of a biomarker strategy for ALS
Amyotrophic lateral sclerosis (ALS) can be broken down into three major underlying pathology components: neurodegeneration, inflammation, and neuromuscular dysfunction. At Precision, we’ve developed a strategy covering diagnostic/predictive and pharmacodynamic/prognostic biomarkers for each of these three disease components. In the hypothetical case shown here, the therapeutic is a gene therapy, and neuron-derived exosomes provide a unique means for assesing target engagement by quantifying the gene product’s neuronal expression level. Epiontis ID, a Precision-developed technology for immunophenotyping, is also useful for assessing whether a therapeutic modulates the altered immune cell profile associated with ALS.
Discover how our neurology and CNS expertise can advance your clinical development program
Discover how our neurology and CNS expertise can advance your clinical development program
Services supporting clinical development of neurology and CNS therapeutics
Clinical Trial Design
Advanced trial-design approaches—including basket, umbrella, and adaptive trials—deliver biomarker-driven clinical research. Deep experience in these highly complex trial designs maximizes both insights and efficiency.
Bioanalytical Testing
Bioanalytical analysis of biologics, cell therapies, gene therapies, and companion diagnostics development from the globally recognized leaders in immunogenicity testing.
Biomarker Data Management
The only platform proven to successfully integrate multiomic data. Flexible pipelines and AI-driven smart mapping are deployed for rapid insights within a clinical trial or at an enterprise level as a consolidated clinical data ecosystem. Collaborative insight generation is empowered via a web-based user interface or through direct access to data via API.