Gene Therapy
Development Services
Gene therapy development is a young field with a wide range of challenges that include a dynamic regulatory environment, pre-existing and treatment-induced immunity to the viral vectors, and even patient recruitment and adherence for gene therapies targeting rare diseases.
Precision’s integrated approach to preclinical and clinical development ensures that the comprehensive services and expertise needed to create solutions for every gene therapy development challenge, from IND-enabling assays to commercialization, are ready to advance your development program.

Optimizing successful development of viral vector gene therapies, gene therapy trials, and companion diagnostics
Delivering evidence at every key gene therapy development stage
Precision delivers leadership in regulatory strategy, specialization in immunogenicity studies and rare disease trials, and the expertise to effectively coordinate both a therapeutic and companion diagnostic strategy in parallel.
Industry-leading expertise in biomarker and immunogenicity assays
Biomarker assays are critical for gene therapy development, especially for evaluating T and B cell immunogenicity.
Precision is at the forefront of immunogenicity testing for gene therapy trials, with thought leaders who are helping to shape the regulatory landscape and standardize methods for AAV-based assays.
Learn more about our specialty lab services >
Genomics expertise to analyze preclinical gene therapy safety
- Viral biodistribution via ddPCR, qPCR (mouse/NHP, or other species as required)
- Assess gene editing accuracy and specificity via NGS (mouse/NHP, or other species as required)
- DNA/RNA extraction via automated systems
Learn more about our genomics services >
Full support for rare disease trial execution
Gene therapies often target small patient populations, presenting unique challenges. We develop and design trials with a patient-centric approach and utilize advanced statistical methods to reach small populations and meet complex endpoints. Our expertise is reflected in a 100% data submission acceptance rate.
Learn more about our rare and orphan disease expertise>
End-to-end companion diagnostics and regulatory process experience
We develop and execute strategies for assays, trials and CDx development in tandem, an approach necessary to ensure rapid time to market. In addition, we offer fully-compliant (CLIA) cell-based NAb assays for CDx and commercial use.
Learn more about our companion diagnostics capabilities>
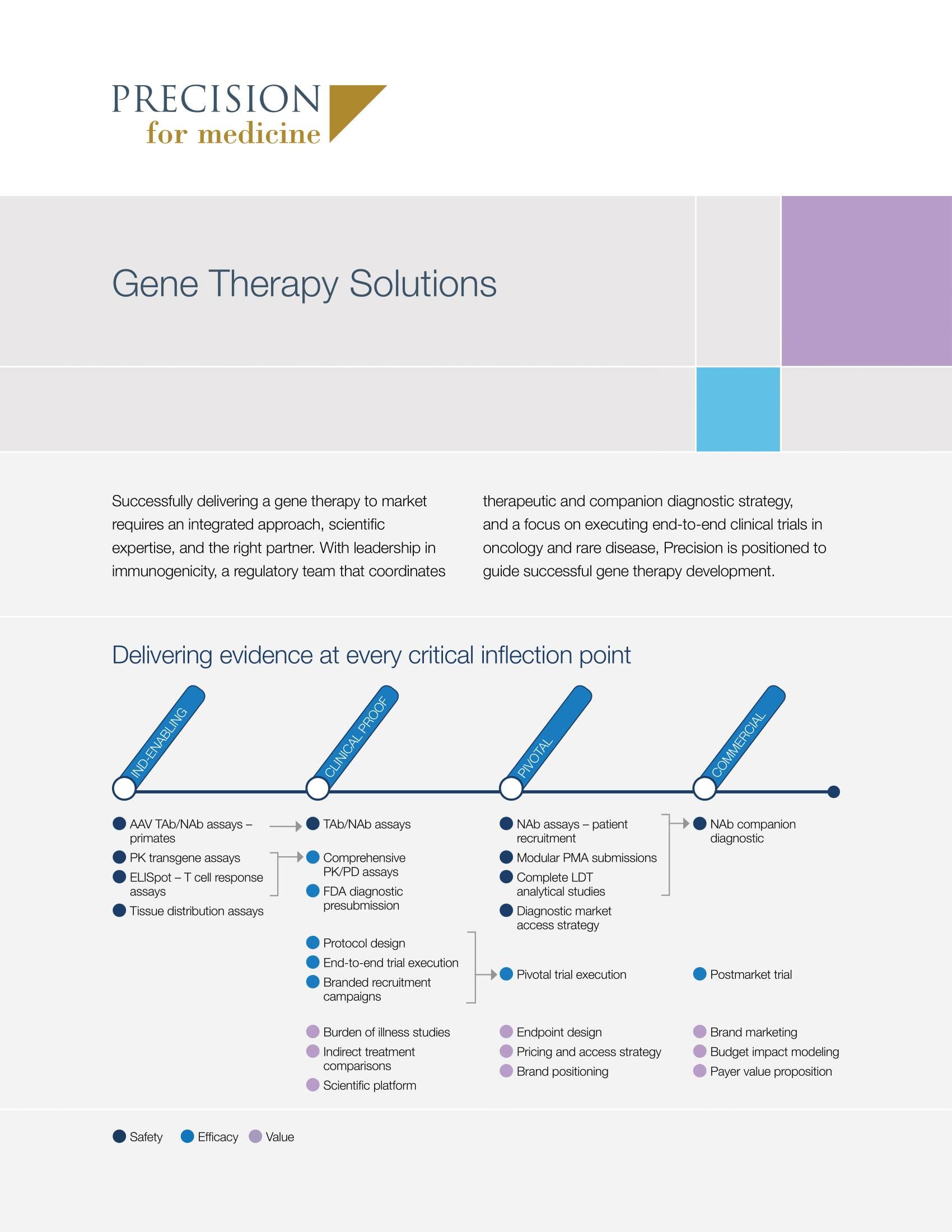
Gene therapy clinical development solutions
Learn more about Precision’s capabilities for accelerating gene therapy development.

Preclinical services for gene therapy, gene editing therapy, and oncolytic virus development
Partner with the experts in cell and gene therapy development, manufacturing, and commercialization
Delivering a cell or gene therapy to market requires a nuanced, comprehensive, and integrated approach. That’s why all Precision clients have access to ADVANCE. The Precision ADVANCE cell and gene therapy collective brings together our unique expertise and
capabilities across the continuum of early development, biological manufacturing and commercialization. These interconnected services support and protect the uninterrupted advance of your therapy.
Discover how our gene therapy experts can advance your clinical development program
Discover how our gene therapy experts can advance your clinical development program
Services that support cell and gene therapy development
Clinical Development Strategy
Tailored strategies consider the scientific, regulatory, and commercial factors that shape each trial, mitigating risk and advancing the development pathway.
Cytokine Analysis
Build a quantitative understanding of the cytokines present in your sample. We leverage a range of assay technologies to maximize data from even challenging samples.
Bioanalytical Testing
Bioanalytical analysis of biologics, cell therapies, gene therapies, and companion diagnostics development from the globally-recognized leaders in immunogenicity testing.
